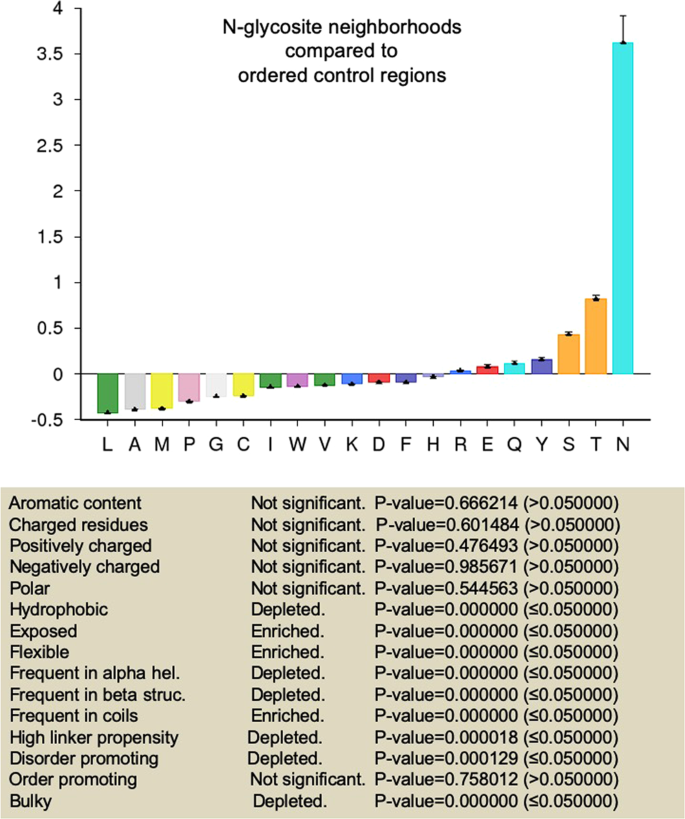
Substitution of Leu-131 with polar amino acids (aspartate and asparagine) also resulted in strongly defective coupling, whereas phenylalanine (found in the equivalent position in the beta 2 adrenoceptor) can replace leucine without losing PI coupling ability of Hm1. Similar amino-terminal domains were identified in the VP16 homologs of other alphaherpesviruses. The position equivalent to Leu-131 in Hm1 contains a bulky hydrophobic amino acid (L, I, V, M, or F) in nearly all cloned GPCRs. Transactivation by the native ORF10 protein was abolished when Phe-28 was replaced by Ala. Extensive mutagenesis of Phe-28 in GAL4-ORF10 fusion proteins demonstrated the importance of an aromatic or bulky hydrophobic amino acid at this position, as shown previously for Phe-442 of VP16. bulky and hydrophobic amino acid residues into the ATCUN motif. However, when amino acids are incorporated into peptide chains, they no longer have amino and carboxy groups, so they’re characterized on the basis of their si.
They all have amino groups and carboxylic acid groups and exist as zwitterions.
#BULKY HYDROPHOBIC AMINO ACIDS SERIES#
With a series of GAL4-ORF10 fusion proteins, we mapped the ORF10 transcriptional-activation domain to the amino-terminal region (aa 5-79). Copper complexes formed by an amino terminal Cu2+- and Ni2+-binding (ATCUN) motif have. Answer: Which amino acids are hydrophilic All of them. These residues are normally located inside the protein core, isolated from solvent. A protein sequence comparison procedure termed hydrophobic cluster analysis was used to identify a motif centered at Phe-28, near the amino terminus of ORF10, that strongly resembles the sequence of the activating domain surrounding Phe-442 of VP16. The hydrophobic amino acids include alanine (Ala, A), valine (Val, V), leucine (Leu, L), isoleucine (Ile, I), proline (Pro, P), phenylalanine (Phe, F) and cysteine (Cys). Varicella-zoster virus open reading frame 10 (ORF10) protein, the homolog of the herpes simplex virus protein VP16, can transactivate immediate-early promoters from both viruses.
